Centre Director | Professor William James
Head of Centre | Dr Sally Cowley
Located in the Sir William Dunn School of Pathology, University of Oxford, we have developed powerful disease models, harnessing human induced Pluripotent Stem Cell technology, to address some of the most challenging questions in medical research.
Current research themes

The macrophage is a key innate immune cell in health and disease, and we have developed very powerful methods for dissecting physiological and pathological pathways in both infectious and non-infectious disease. We are currently focusing on models of brain and lung macrophages.
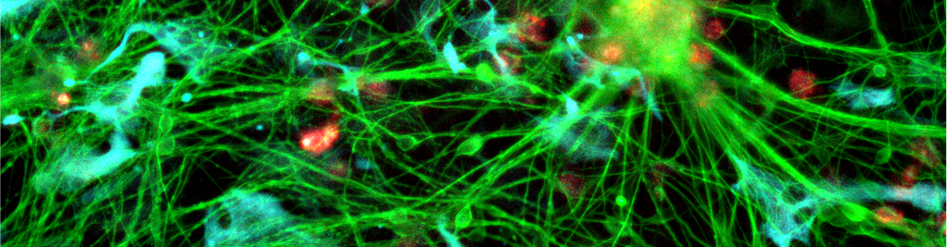
The resident macrophages of the brain parenchyma – microglia – are critical in maintaining brain health, and when they are dysfunctional, diseases from dementias to neurocognitive disorders can result. Several of the genes associated with Alzheimer’s disease are specifically expressed in microglia rather than other brain cell types, and progression of the disease is characterised by failure of tissue homeostasis by microglia, with accumulation of misfolded amyloid beta and tau proteins.
In respiratory infections, such as SARS-CoV-2, polarized epithelial cells of the upper and lower respiratory tract are often the primary host cells. They can interact with local lung macrophages, either limiting viral spread and resolving infection, or inducing recruitment of infiltrating inflammatory immune cells, resulting in tissue damage and life-threatening disease.
To dissect the molecular pathways employed by tissue macrophages and their neighbouring cells in maintaining tissue integrity, and to understand the processes by which pathogens, inherited differences and environmental variables can disrupt them to cause disease, we employ human induced Pluripotent Stem Cell technology. We have developed cutting-edge methods for generating and genetically modifying iPSC and differentiating them to macrophages, microglia and neighbouring cell types. These experimentally tractable and physiologically authentic “disease in a dish” models put us at the forefront of studying these important human disease processes.