The James & Lillian Martin Centre at the Sir William Dunn School of Oxford oversees the core Containment level 3 Facility that supports work of the Centre and many collaborating groups, on SARS-CoV-2 and HIV-1. The following are some research publications that have resulted from this work:
Immune responses to SARS-CoV-2 infection and vaccines
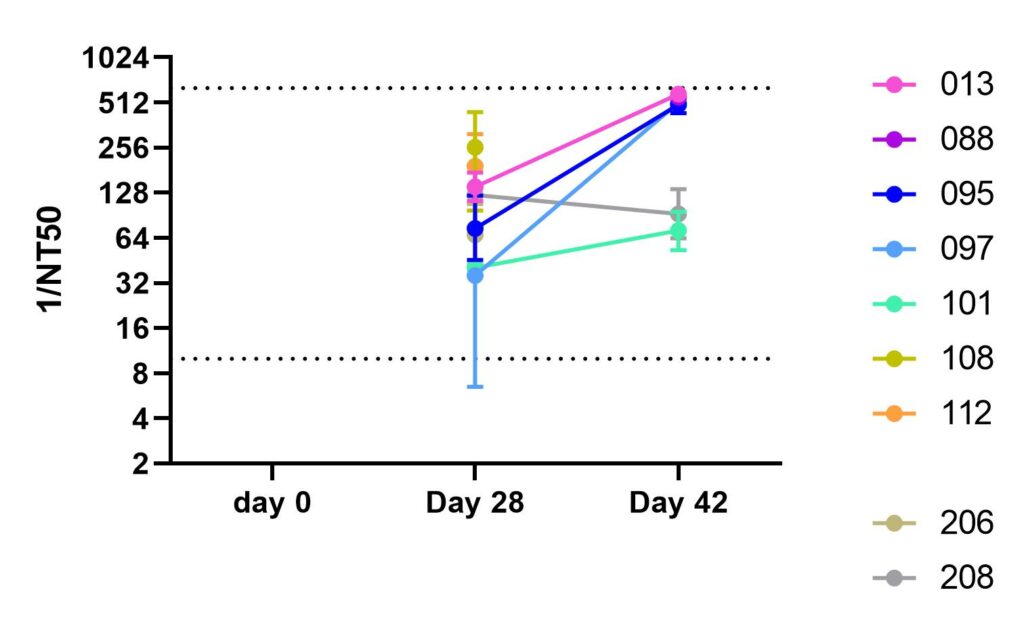
Skelly, D.T., A.C. Harding, J. Gilbert-Jaramillo, M.L. Knight, S. Longet, A. Brown, S. Adele, et al. “Two Doses of SARS-CoV-2 Vaccination Induce Robust Immune Responses to Emerging SARS-CoV-2 Variants of Concern.” Nature Communications 12, no. 1 (2021). https://doi.org/10.1038/s41467-021-25167-5.
Folegatti, Pedro M, Katie J Ewer, Parvinder Aley, Brian Angus, Stephan Becker, Sandra Belij-Rammerstorfer, Duncan Bellamy, et al. “Safety and Immunogenicity of the ChAdOx1 NCoV-19 Vaccine against SARS-CoV-2: A Preliminary Report of a Phase 1/2, Single-Blind, Randomised Controlled Trial.” The Lancet 396, no. 10249 (July 20, 2020): 467–78. https://doi.org/https://doi.org/10.1016/S0140-6736(20)31604-4.
Peng, Y., S.L. Felce, D. Dong, F. Penkava, A.J. Mentzer, X. Yao, G. Liu, et al. “An Immunodominant NP<inf>105–113</Inf>-B*07:02 Cytotoxic T Cell Response Controls Viral Replication and Is Associated with Less Severe COVID-19 Disease.” Nature Immunology 23, no. 1 (2022): 50–61. https://doi.org/10.1038/s41590-021-01084-z.
Huang, Kuan Ying A., Tiong Kit Tan, Ting Hua Chen, Chung Guei Huang, Ruth Harvey, Saira Hussain, Cheng Pin Chen, et al. “Breadth and Function of Antibody Response to Acute SARS-CoV-2 Infection in Humans.” Edited by Benhur Lee. PLoS Pathogens 17, no. 2 (February 26, 2021): e1009352. https://doi.org/10.1371/JOURNAL.PPAT.1009352.
Zhou, Daming, Wanwisa Dejnirattisai, Piyada Supasa, Chang Liu, Alexander J. Mentzer, Helen M. Ginn, Yuguang Zhao, et al. “Evidence of Escape of SARS-CoV-2 Variant B.1.351 from Natural and Vaccine Induced Sera.” Cell, February 23, 2021. https://doi.org/10.1016/j.cell.2021.02.037.
Huo, Jiandong, Halina Mikolajek, Audrey Le Bas, Jordan J. Clark, Parul Sharma, Anja Kipar, Joshua Dormon, et al. “A Potent SARS-CoV-2 Neutralising Nanobody Shows Therapeutic Efficacy in the Syrian Golden Hamster Model of COVID-19.” Nature Communications 2021 12:1 12, no. 1 (September 22, 2021): 1–18. https://doi.org/10.1038/s41467-021-25480-z.
Supasa, P., D. Zhou, W. Dejnirattisai, C. Liu, A.J. Mentzer, H.M. Ginn, Y. Zhao, et al. “Reduced Neutralization of SARS-CoV-2 B.1.1.7 Variant by Convalescent and Vaccine Sera.” Cell 184, no. 8 (2021): 2201-2211.e7. https://doi.org/10.1016/j.cell.2021.02.033.
Zhou, D., H.M.E. Duyvesteyn, C.-P. Chen, C.-G. Huang, T.-H. Chen, S.-R. Shih, Y.-C. Lin, et al. “Structural Basis for the Neutralization of SARS-CoV-2 by an Antibody from a Convalescent Patient.” Nature Structural and Molecular Biology 27, no. 10 (2020): 950–58. https://doi.org/10.1038/s41594-020-0480-y.
Mistry, P., F. Barmania, J. Mellet, K. Peta, A. Strydom, I.M. Viljoen, W. James, S. Gordon, and M.S. Pepper. “SARS-CoV-2 Variants, Vaccines, and Host Immunity.” Frontiers in Immunology 12 (2022). https://doi.org/10.3389/fimmu.2021.809244.
Development of SARS-CoV-2-neutralizing Monoclonal antibodies, nanobodies and chimeric proteins
Huang, K.-Y.A., D. Zhou, T.K. Tan, C. Chen, H.M.E. Duyvesteyn, Y. Zhao, H.M. Ginn, et al. “Structures and Therapeutic Potential of Anti-RBD Human Monoclonal Antibodies against SARS-CoV-2.” Theranostics 27, no. 1 (2022): 1–17. https://doi.org/10.7150/THNO.65563.
Qiang, M., P. Ma, Y. Li, H. Liu, A. Harding, C. Min, F. Wang, et al. “Neutralizing Antibodies to SARS-CoV-2 Selected from a Human Antibody Library Constructed Decades Ago.” Advanced Science 9, no. 1 (2022). https://doi.org/10.1002/advs.202102181.
Girt, G.C., A. Lakshminarayanan, J. Huo, J. Dormon, C. Norman, B. Afrough, A. Harding, W. James, R.J. Owens, and J.H. Naismith. “The Use of Nanobodies in a Sensitive ELISA Test for SARS-CoV-2 Spike 1 Protein.” Royal Society Open Science 8, no. 9 (2021). https://doi.org/10.1098/rsos.211016.
Huo, Jiangdong, Audrey Le Bas, Reinis R. Ruza, Helen M.E. Duyvesteyn, Halina Mikolajek, Tomas Malinauskas, Tiong Kit Tan, et al. “Neutralizing Nanobodies Bind SARS-CoV-2 Spike RBD and Block Interaction with ACE2.” Nature Structural and Molecular Biology, July 13, 2020, 1–9. https://doi.org/10.1038/s41594-020-0469-6.
Miao, X., Y. Luo, X. Huang, S.M.Y. Lee, Z. Yuan, Y. Tang, L. Chen, et al. “A Novel Biparatopic Hybrid Antibody-ACE2 Fusion That Blocks SARS-CoV-2 Infection: Implications for Therapy.” MAbs 12, no. 1 (2020). https://doi.org/10.1080/19420862.2020.1804241.
Molecular Cell Biology of SARS-CoV-2
Zhang, Peijun. “Correlative Multi-Scale Cryo-Imaging Unveils SARS-CoV-2 Assembly and Egress,” January 19, 2021. https://doi.org/10.21203/rs.3.rs-134794/v1.
Lee, J.Y., P.A.C. Wing, D.S. Gala, M. Noerenberg, A.I. Järvelin, J. Titlow, X. Zhuang, et al. “Absolute Quantitation of Individual SARS-CoV-2 RNA Molecules Provides a New Paradigm for Infection Dynamics and Variant Differences.” ELife 11 (2022). https://doi.org/10.7554/eLife.74153.
Wing, Peter A.C., Thomas P. Keeley, Xiaodong Zhuang, Jeffrey Y. Lee, Maria Prange-Barczynska, Senko Tsukuda, Sophie B. Morgan, et al. “Hypoxic and Pharmacological Activation of HIF Inhibits SARS-CoV-2 Infection of Lung Epithelial Cells.” Cell Reports 35, no. 3 (2021). https://doi.org/10.1016/j.celrep.2021.109020.
Dejnirattisai, W., D. Zhou, H.M. Ginn, H.M.E. Duyvesteyn, P. Supasa, J.B. Case, Y. Zhao, et al. “The Antigenic Anatomy of SARS-CoV-2 Receptor Binding Domain.” Cell 184, no. 8 (2021): 2183-2200.e22. https://doi.org/10.1016/j.cell.2021.02.032.
Matthews, P.C., M.I. Andersson, C.V. Arancibia-Carcamo, K. Auckland, J.K. Baillie, E. Barnes, T. Beneke, et al. “SARS-CoV-2 RNA Detected in Blood Products from Patients with COVID-19 Is Not Associated with Infectious Virus.” Wellcome Open Research 5 (2020). https://doi.org/10.12688/wellcomeopenres.16002.2.