anne.hedegaard@path.ox.ac.uk
PubMed Publications
ORCiD
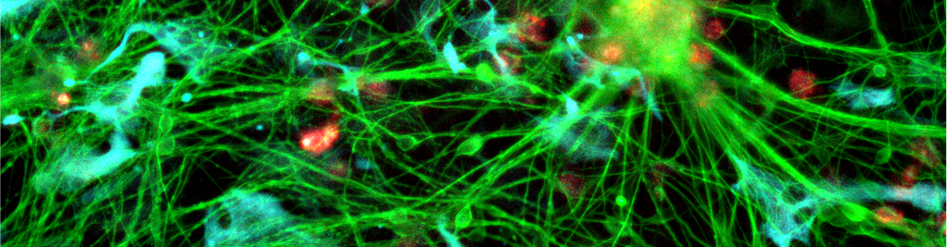
I joined the lab in 2020 on a project in collaboration with, and funded by, Eli Lilly and Company. I started off investigating how inflammatory signals associated with neurodegeneration, and Alzheimer’s Disease (AD) in particular, propagate from one brain cell type to another. For this purpose, I was interested in establishing more physiologically relevant in vitro models of human brain cells and used induced pluripotent stem cell (iPSC) technology to derive both neurons, astrocytes and microglia, which can be studied both on their own and in co-culture to elucidate their cross-talk.
More recently, I have been working on understanding the apparent connection between the AD risk-factor gene BIN1, and the release of the disease-relevant protein tau by microglia. BIN1 is the second highest genetic risk factor for sporadic AD, but exactly how it conveys this risk is not entirely clear. However, lowering the expression of BIN1 has been shown to lower the amount of tau associated with extracellular vesicles released by microglia. This could be important, as pathological forms of tau can propagate between brain cells and cause normal tau in a healthy cell to aggregate, thereby spreading the disease throughout the brain. We think microglia is an important cell-type in this propagation, as part of their normal “job” involves clearing up aggregated proteins, like tau, but they might be unable to fully degrade it, and end up disposing of tau in extracellular vesicles.
As part of the Partners in Tau collaboration with UC Santa Barbara, I am exploring how manipulating the level of BIN1 in iPSC-derived microglia affects their processing and release of tau.
I find that being high up and upside-down helps get ideas flowing, so I achieve this outside the lab through acrobatics and by performing on an aerial hoop or trapeze on a regular basis.